|
Protons have a positive charge and electrons a negative charge. Protons and electrons have equal and opposite charges. Because the number of neutrons in an atom can vary, there can be several different atomic weights for most elements. For most purposes, the atomic weight can be thought of as the number of protons plus the number of neutrons. Protons and neutrons make up the nucleus of an atom and the electrons orbit.Įlectrons contribute only a tiny part to the mass of the atomic structure, however, they play an important role in the chemical reactions that create molecules. The atomic mass or weight is measured in atomic mass units. The total mass of an atom, including the protons, neutrons and electrons, is the atomic mass or atomic weight. The mass of a proton or neutron increases when the particle attains extreme speed, for example in a cyclotron or linear accelerator. A neutron is electrically neutral and has a rest mass, denoted m n, of approximately 1.675 x 10 -27.A proton has a rest mass, denoted m p, of approximately 1.673 x 10 -27 kilogram (kg). For example, sodium has 11 protons, and its atomic number is 11. Different elements' atomic numbers are found in the Periodic Table of Elements. 
The number of protons in the nucleus of an atom is the atomic number for the chemical element. 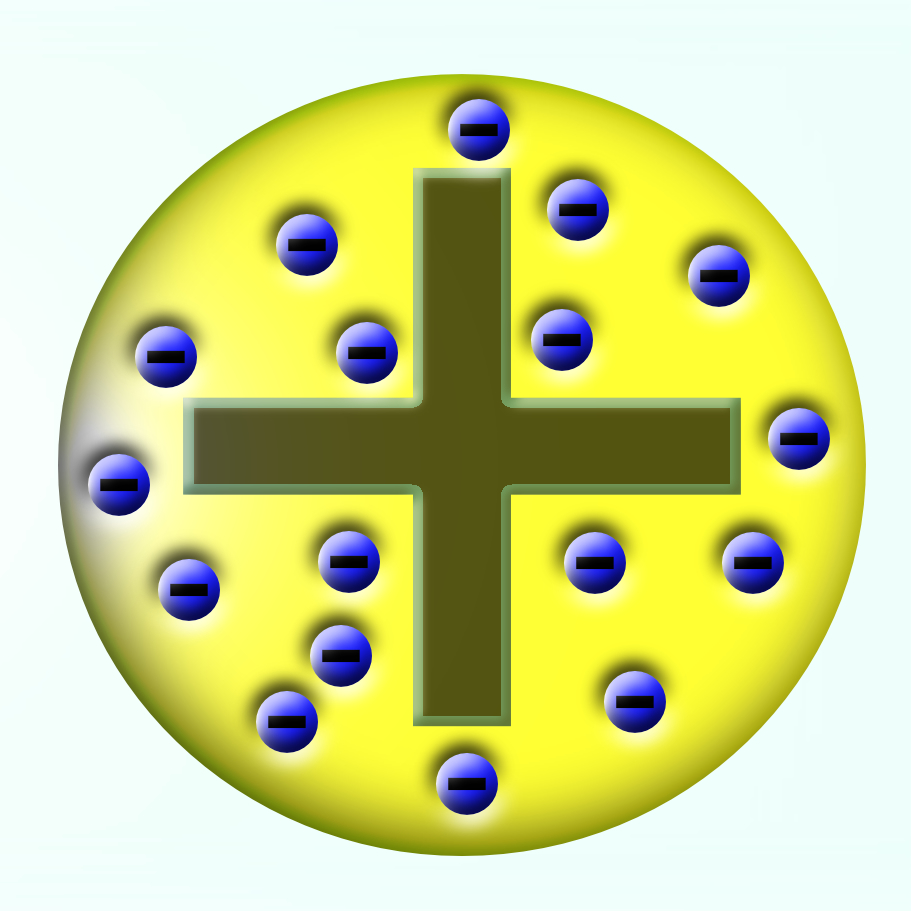
Protons and neutrons are subatomic particles that make up the center of the atom, or its atomic nucleus. Anything that takes up space and anything with mass is made up of atoms. The nucleus is positively charged and contains one or more relatively heavy particles known as protons and neutrons.Ītoms are the basic building blocks of matter. An atom consists of a central nucleus that is surrounded by one or more negatively charged electrons. An atom is a particle of matter that uniquely defines a chemical element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
